
Finding Research Collaborators : Ultimate Guide 2026
Research collaboration is one of the fastest ways to move from an idea to real clinical proof. But in healthcare, “finding a collaborator” is not the same as “hiring help.” The right person can unlock patients, approvals, and trial execution. The wrong person can stall you for months.
What Is Collaborative Research?
Collaborative research is when two or more people or institutions share effort, data, and expertise to answer a research question. It works because each person brings something the others cannot easily copy:
- Patients
- Methods
- Datasets
- Operational muscle
It also tends to get more visibility. A 2024 bibliometric study in Research Policy found that papers with international co-authors consistently receive higher citation counts than single-country papers because collaboration exposes work to multiple research networks. For founders, collaboration is not just about publishing. [ Source ]
It also:
- Builds clinical credibility
- Creates trial pathways
- Provides real-world feedback before spending large product budgets
-
In a startup, you usually hire people to execute specific tasks. In clinical research, however, collaborators play a very different role. They are often gatekeepers to execution itself. A trial-ready clinician does not just contribute effort; they can unlock patient access and referral pathways, provide access to site infrastructure such as coordinators, systems, and workflows, help navigate institutional sign-off and IRB readiness, and bring practical trial execution experience. That is why collaboration in healthcare looks much more like shared leadership than simple task-based support. It is also why reputation alone is not enough. A well-known name may add credibility, but execution history is often the stronger signal of whether a collaborator can actually help move a study forward.
Myths to Ignore Before You Start
Most delays in finding research collaborators come from avoidable assumptions. Many founders and researchers approach collaboration with ideas that sound reasonable but do not reflect how clinical research actually works. When these assumptions guide your search, you can spend months speaking with the wrong people or asking for the wrong type of help. Addressing these myths early makes the search process much faster and more predictable.
Myth 1: The Most Famous Researcher Is the Best Partner
One common assumption is that the most famous researcher in a field will automatically be the best collaborator. In practice, fame often comes with limited bandwidth. Highly visible researchers may have multiple projects, administrative responsibilities, and speaking commitments, which means they may not be deeply involved in day-to-day clinical work. They may also be removed from the operational realities of patient recruitment and study coordination.
In many situations, mid-career clinicians are stronger long-term collaborators. These clinicians typically still see a high volume of patients, actively run ongoing studies, understand real clinical workflows, and can engage in the operational details required to execute a study successfully. If your goal is to run a clinical trial, it is usually better to prioritize execution capability and patient access rather than prestige alone.
Myth 2: A Great Publication Record Means They Can Run a Study
Another common misconception is that a strong publication record guarantees strong trial execution ability. Publishing and running a clinical study require very different skill sets. Some researchers are exceptional at data analysis, writing manuscripts, and developing theoretical frameworks but may not be involved in patient recruitment, study logistics, or regulatory coordination.
If your goal is to run a clinical study, it is important to verify operational experience. This includes checking whether the investigator has served as a principal investigator (PI) or sub-investigator (Sub-I), whether their institution has the infrastructure required to run trials, and whether they have a history of recruiting the relevant patient population. Trial registries such as ClinicalTrials.gov are often the fastest way to verify this type of experience.
Myth 3: Cold Outreach Is Enough
Many people assume that sending cold emails will be sufficient if the idea is strong enough. In reality, even good ideas often get lost in busy inboxes. Clinical research is heavily network-driven, and trust typically develops through professional relationships and shared context.
Cold outreach can still work, but warm introductions are usually much more effective. An analysis of 12 million outreach emails found that personalized emails receive approximately 32.7% higher response rates, while generic outreach messages receive replies only about 8.5% of the time. The practical takeaway is simple: when reaching out to potential collaborators, reference their work specifically, demonstrate why your project aligns with their interests, and propose a small, low-commitment next step such as a short exploratory conversation. [Source]
Why Finding Research Collaborators Is Difficult
Many people assume that research collaboration fails because the science itself is difficult. In reality, the biggest obstacles are rarely technical. The real barriers are usually time, trust, and risk. Clinicians and researchers operate in environments where schedules are tight, reputations matter, and poorly structured collaborations can create significant professional problems. Understanding these constraints helps you design collaboration requests that are realistic and respectful of how research actually works.
The Hidden Constraints: Time, Trust, and Risk
Clinicians often ignore collaboration requests not because they are uninterested, but because their time and professional reputation are constantly under pressure. Many physicians balance clinic schedules, teaching responsibilities, administrative duties, and multiple ongoing studies. Even when a project sounds interesting, the practical reality of adding another responsibility can make it difficult to engage.
Trust is another major factor. Researchers are cautious when approached by unknown partners, especially when details about data handling, study goals, or institutional processes are unclear. If the intent of a collaboration is not well explained, it can raise concerns about professionalism, scientific quality, or compliance.
Risk also plays an important role. Collaborations can carry reputational and ethical consequences if the project is poorly designed or conducted. A survey of 6,673 researchers found that 46.6% reported authorship conflicts during collaborations, while 24.6% experienced hostility within teams and 16.4% reported colleagues undermining their work. These experiences make many investigators cautious about entering new partnerships, particularly when they have previously encountered difficult collaborations. [Source]
Misaligned Incentives
Even when both sides are well intentioned, founders and academic investigators often approach collaboration with different priorities. Founders typically focus on speed, feasibility validation, regulatory clarity, and generating evidence that supports fundraising or product development. Their primary goal is often to learn quickly and move toward implementation.
Investigators, however, are usually driven by different incentives. Academic careers often depend on publications, grant funding, career progression, and leadership within research programs. As a result, the questions they prioritize may be shaped by what leads to high-quality academic output rather than rapid product validation.
These differences can create friction if they are not acknowledged early. A global survey of more than 9,600 academics found that 69% consider a shared research approach the most important factor before collaborating, and 44% prioritize shared ethics and values. Aligning expectations about goals, timelines, and outcomes early in the conversation can prevent many of the conflicts that emerge later in a project. [Source]
Institutional Capability Is Often the Real Bottleneck
A willing clinician does not automatically mean a capable research site. Even highly motivated investigators may struggle to run studies if their institution lacks the infrastructure needed to support clinical research. Operational factors often determine whether a collaboration can actually move forward.
For example, the presence of a dedicated research coordinator can significantly affect a study’s feasibility, since coordinators handle tasks such as patient recruitment, consent processes, and data management. Institutional review board (IRB) timelines also matter, as approval processes can vary widely across hospitals and universities. Sites that have recently conducted similar trials are usually more prepared to handle new studies because they already have established processes and trained staff.
Data systems are another important consideration. Hospitals with structured research databases, EHR extraction capabilities, or tools such as REDCap can support data collection much more efficiently. In addition, contract management and compliance teams are often required to handle agreements, regulatory documentation, and financial arrangements. Without these operational systems in place, even experienced clinicians may struggle to execute a study successfully.
Know What You Need Before Searching
A successful collaborator search begins with clarity about what you actually need. Many outreach efforts fail because the request itself is vague. If the research question, study design, or expected roles are unclear, potential collaborators may hesitate to engage.
Defining the job before searching for the person makes the process far more effective. When you clearly understand the goal of the collaboration and the expertise required, it becomes much easier to identify the right people and communicate your needs succinctly.
Clarify Your Collaboration Goal
Different collaboration goals require different types of partners. One common mistake is trying to accomplish multiple objectives simultaneously, which can create confusion about responsibilities and expectations.
If the goal is to publish academic work, the most valuable collaborators are often method experts, experienced statisticians, or senior authors who can strengthen the study design and help guide the writing process. In these cases, projects may involve retrospective analyses or observational studies that lead to manuscripts.
If the goal is validation, the best collaborators are active clinicians who treat the relevant patient population. These partners can help evaluate whether the study question reflects real clinical workflows and whether the proposed endpoints make sense in practice.
If the goal is to run a clinical trial, execution experience becomes the most important factor. In this case, collaborators should include principal investigators or sub-investigators who have previously conducted trials and work at sites with research coordinators and operational support. A pilot clinical study is often the first step in this type of collaboration.
Define the Collaboration Gap
Before reaching out to potential collaborators, it is useful to prepare a simple internal worksheet that clarifies what the project requires. This worksheet should include the core research question, the target population, the clinical or research setting, and the methodological expertise needed to execute the study. It should also outline what type of data access is required, the specific role you expect the collaborator to play, and the approximate time commitment expected during the early stages of the project.
Another important consideration is decision authority. Clarifying who will make key decisions about study design, protocol changes, and analysis plans can prevent confusion later. If these elements are not clearly defined internally, outreach conversations may feel unfocused or premature.
Decide What Value You Offer
Research collaboration is fundamentally a value exchange rather than a favor. Investigators are much more likely to engage when the collaboration clearly advances their goals or reduces their workload. Offering something concrete demonstrates that you understand the realities of research work.
Value can take several forms. Providing a draft protocol or research aims can save collaborators significant time by allowing them to react to an existing plan rather than build one from scratch. Technical contributions such as statistical analysis, engineering support, or data pipelines can also make a project more attractive. In some cases, financial support for study costs or coordinator time can make the collaboration feasible for a clinical site.
Other forms of value include tools that improve data collection, opportunities for authorship on resulting publications, or the ability to launch a small pilot study that generates early evidence. Platforms like StudyConnect can also help structure early conversations by making it easier for founders and clinicians to discuss feasibility, study design, and collaboration expectations before significant resources are invested.

Where Researchers Traditionally Find Collaborators
Most research collaborations begin through existing academic or professional context rather than random outreach. Researchers often discover potential collaborators through papers they read, talks they attend, colleagues they already know, or datasets that multiple groups are working with. These environments provide strong signals of alignment because they show who is actively working on similar questions. As a result, they remain some of the most reliable channels for identifying collaborators.
Authors of Papers You Read
One of the fastest ways to identify collaborators is by studying the authors of papers that are directly related to your research question. A strong paper can effectively become a map of the research network working in that area. Start by identifying three to five highly relevant papers and reviewing the full list of authors on each. Within these lists, prioritize the corresponding author, the first author, and the senior author, as these individuals are often the most actively involved in leading the research. From there, explore other papers produced by the same group and begin building a shortlist of roughly twenty researchers who are consistently publishing in that space. This approach quickly reveals the active labs and investigators shaping the field.
Conferences and Seminars
Academic conferences and seminars provide valuable opportunities for building collaboration through warm introductions rather than cold outreach. Talks and presentations reveal who is actively working on specific problems, and they allow researchers to ask questions and start conversations in a natural context. Effective preparation can significantly improve the value of these interactions. Reviewing speaker lists in advance helps identify researchers who are most relevant to your work, while attending sessions aligned with your topic increases the likelihood of meaningful discussions. Asking thoughtful questions during Q&A sessions and following up with speakers within a day or two can often lead to early-stage collaboration conversations. Even in highly digital research environments, conference networking remains one of the strongest channels for forming research partnerships.
Citation Networks
Another powerful method for discovering collaborators is through citation networks. Researchers who cite similar work are often investigating closely related problems, making them strong potential collaborators. A simple way to use this method is to begin with a key paper that closely matches your research topic. By using the “Cited by” feature in Google Scholar, you can view newer papers that reference the original work. Scanning these papers reveals additional authors who are actively contributing to the same area of research. Adding these researchers to a shortlist for outreach often produces higher-quality collaboration opportunities because the intellectual alignment already exists.
What Is a Citation Trail?
A citation trail refers to the chain of research papers that reference one another over time. Instead of guessing who might be interested in your project, you follow the trail of citations to identify researchers who have already shown interest in the same topic. This approach provides a clear context for outreach because you can reference the shared research connection directly. For example, a message might mention that the researcher cited a specific paper and that your project builds on a similar approach in a different setting. Citation trails often reveal active mid-career researchers who are deeply engaged in current work and are more likely to participate in practical collaborations.
How to Find Research Collaborators: Step-by-Step Workflow
Finding collaborators becomes much easier when you follow a repeatable process instead of searching randomly. A structured workflow helps you consistently identify relevant researchers, evaluate their work, and build a focused outreach list.
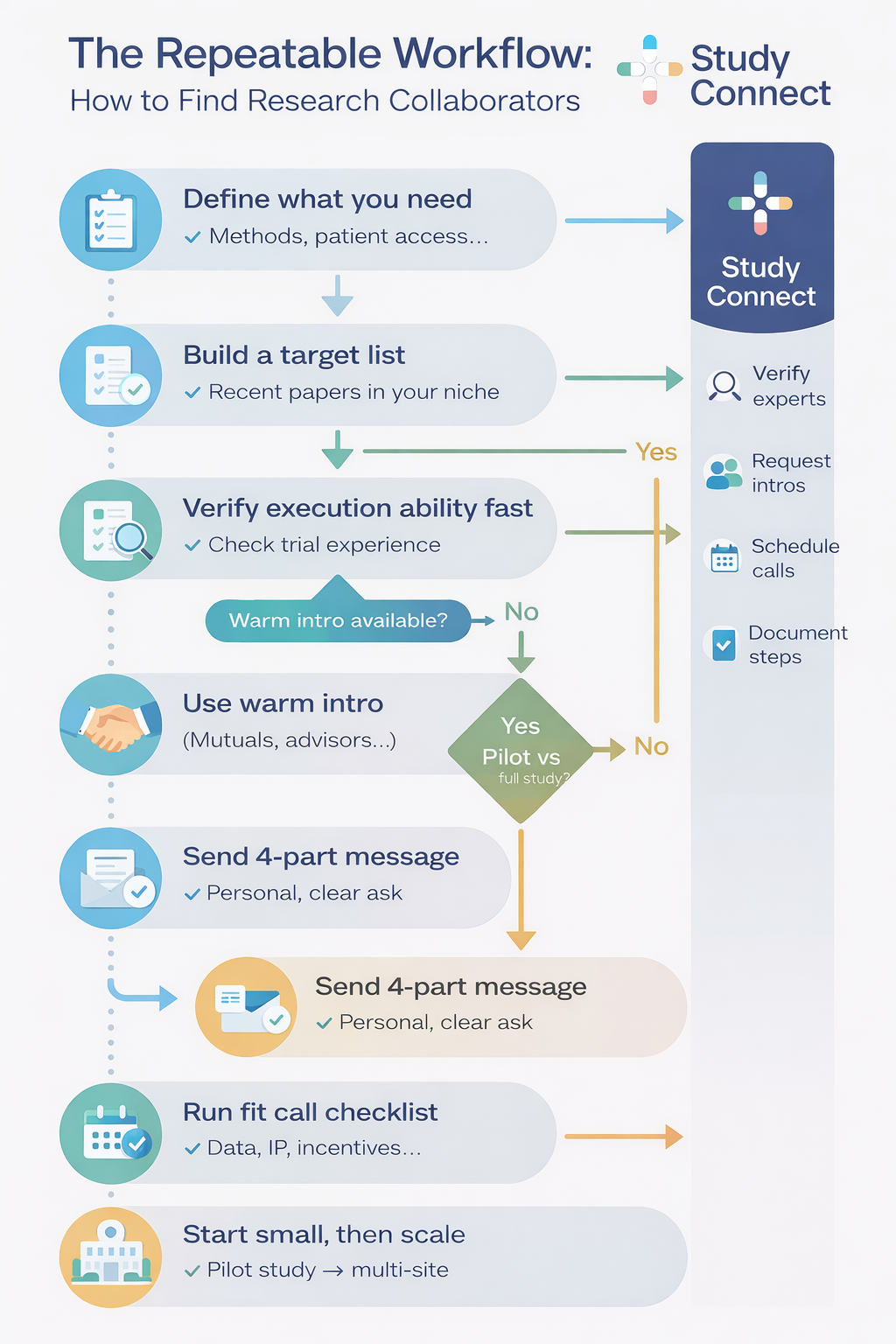
Step 1: Build a Keyword Map
The first step is to create a small keyword map that describes your research niche. These keywords typically combine four elements: the condition being studied, the research method, the outcome of interest, and the clinical setting.
For example, a phrase like “atrial fibrillation wearable outpatient monitoring” combines all four elements into a precise search query. Similar phrases might include “COPD remote monitoring cohort,” “heart failure readmission EHR study,” or “oncology real-world registry study.” This keyword map becomes a reusable tool that can be applied across multiple research platforms when searching for collaborators.
Step 2: Use Google Scholar
Google Scholar is one of the most efficient tools for identifying active researchers in a specific field. By searching your keyword phrases, you can quickly locate recent papers and identify the authors behind them.
Reviewing author profiles reveals their publication history, their co-author networks, and the institutions they are affiliated with. Over time, this process helps you recognize clusters of researchers who frequently work together. Creating alerts for important keyword phrases also allows you to track new publications automatically and stay aware of emerging work in your field.
Step 3: Use ResearchGate
ResearchGate provides another useful channel for engaging with researchers, particularly for lightweight conversations around published work. Instead of sending generic collaboration requests, a more effective approach is to engage with specific aspects of a paper.
Requesting the full text of a study, asking a targeted methodological question, or discussing a particular dataset can start a meaningful exchange. If the conversation develops naturally, suggesting a short exploratory call can open the door to deeper collaboration. Messages that are specific and respectful tend to perform far better than mass outreach or long product-focused introductions.
Step 4: Use Citation Databases
Citation databases such as Web of Science provide additional signals about influential work within a field. These platforms allow you to search for research topics and then sort results by citation counts, helping identify papers that have had a significant impact.
By examining the authors behind highly cited papers, you can identify influential researchers and active research groups. Following the citation trail from these papers often reveals connected teams working on similar problems. While citation metrics highlight academic influence, it is still important to verify whether potential collaborators have practical execution experience, such as participating in clinical trials or managing research sites.
StudyConnect can make the process of finding research collaborators significantly easier by connecting founders and research teams with experienced clinicians and domain experts.
If you are unsure where to start, you can reach out to the StudyConnect team and we can guide you through the entire process- from identifying the right type of experts you should speak with, to understanding potential regulatory hurdles your product may face in clinical settings.
We can also help you gather meaningful feedback from researchers on whether your solution fits real clinical workflows and whether it is likely to be adopted in practice. In addition, StudyConnect hosts a growing network of verified experts and registered researchers across different medical fields.
Through the platform, you can explore relevant profiles, discover specialists working in your area, and book direct calls with them to discuss feasibility, study design, and real-world implementation.
Reach out at : hello@studyconnect.org or register yourself here we will get back to you www.studyconnect.info/waitlist.
How to Evaluate Fit Quickly
You do not need multiple meetings to assess whether a collaborator is a good fit. A few operational questions can quickly reveal whether someone can actually execute a study. For medical startup founders, the goal is not just to find someone interested in your idea, but someone who can realistically help run research in a clinical setting.
Execution Capability
Start by understanding whether the investigator has real trial experience. Three questions usually reveal this quickly: have they served as a principal investigator or sub-investigator before, how many patients with the relevant condition they see each month, and whether they have access to a research coordinator. These answers help distinguish academic interest from operational capability. You can verify them through ClinicalTrials.gov, institutional research pages, or their previous study history. Investigators who regularly run trials will usually talk in terms of recruitment workflows, coordinator support, and timelines rather than only publications.
Patient Access
Patient access is one of the strongest predictors of study success. A site may have strong academic credentials but still struggle to recruit if the relevant patient population is limited. Founders should understand the site’s patient volume, referral pathways, demographic diversity, and past enrollment performance. If the recruitment pathway is unclear, delays are likely later.
Incentive Alignment
Even capable collaborators can run into problems if incentives are not aligned. Early conversations should clarify roles, decision authority, authorship expectations, data ownership, intellectual property boundaries, and publication timelines. These discussions matter because collaboration disputes are common, especially around authorship and ownership. A short written agreement early in the relationship can prevent misunderstandings later.
What Is a Trial-Ready Site?
A trial-ready site is not simply a hospital with interested clinicians. It is an institution with the infrastructure needed to run research efficiently. Four factors usually determine readiness: dedicated research staff such as coordinators or study nurses, a clear IRB approval process, data systems like EHR extraction or REDCap, and a track record of similar studies. Even highly skilled clinicians may struggle if the surrounding institution lacks these capabilities.
How to Approach Collaborators
The goal of your first message is not to secure the collaboration immediately. It is to start a conversation. Busy clinicians receive many vague requests, so outreach should be short, relevant, and easy to respond to.
Warm Introductions
Warm introductions are far more effective than cold outreach. Advisors, co-authors, conference contacts, and clinical champions often provide the best entry points. One good clinician frequently leads to several additional introductions, which is how strong research networks form.
Best First Message Structure
A good first message has four parts: a short reference to the researcher’s work, a brief introduction, a clear collaboration idea, and a low-commitment next step such as a short call. A one-page study brief can help if it explains the research question, target population, and the type of input you are seeking.
Best Practices for Successful Collaboration
Strong research partnerships rely on clarity and consistent communication rather than enthusiasm alone. Roles should be clearly defined, expectations written down, and the team should follow a predictable working rhythm.
Start With Small Studies
Starting small is often the safest path. Feasibility studies, pilot studies, or retrospective analyses allow both sides to test the collaboration before committing to a full clinical trial. These early studies reduce risk, uncover operational challenges, and generate initial evidence that can guide larger trials later.
Feasibility vs Pilot vs Retrospective Studies
A feasibility study evaluates whether a study can realistically be conducted, focusing on recruitment, workflows, and data collection. A pilot study runs a small version of the intended trial to refine protocols and estimate early outcomes. A retrospective study analyzes existing datasets such as EHR records or registries and is often the fastest way to generate early evidence.
Write a Collaboration Agreement
Once a collaboration becomes serious, it helps to document expectations in writing. A basic agreement should define roles, milestones, meeting cadence, authorship rules, data access, confidentiality, and intellectual property boundaries. Clear documentation reduces confusion and protects the relationship if questions arise later.
Operational Rhythm
Consistent structure keeps collaborations productive. Most teams benefit from regular check-ins, a shared milestone document, and a central folder for protocol drafts, data dictionaries, IRB materials, and analysis plans. Clear ownership of tasks ensures that progress does not stall.
Common Collaboration Mistakes
Many collaborations fail due to communication problems rather than scientific issues. Being specific, concise, and realistic can prevent many of these problems.
Vague Requests
Requests like “would love to collaborate” create unnecessary friction. Instead, describe the specific problem you are working on, the role you are proposing, and the next step you would like to take.
Overly Long Messages
Clinicians typically skim emails quickly. Effective outreach usually follows a simple structure: one line explaining why you are contacting them, one line describing the problem, one line outlining the collaboration idea, and one line suggesting a next step.
Choosing Prestige Over Execution
Reputation can help with visibility, but successful trials depend on execution. Prioritize investigators with trial experience, access to the right patient population, coordinator support, and institutions capable of running studies.
How StudyConnect Helps You Build Research Collaborations
Technology has made it easier to find researchers online, but identifying the right collaborators—those who can actually help run studies and validate products—remains difficult. StudyConnect is designed to solve this problem by helping medical startup founders and research teams connect with clinicians and experts who have real operational experience. Instead of navigating large marketplaces with limited context, StudyConnect focuses on verified experts and meaningful conversations that move research and product validation forward.
Verified Expert Matching
Clinical research collaboration requires more than subject knowledge. Founders often need experts who understand patient access, regulatory requirements, institutional workflows, and trial execution. StudyConnect helps you discover clinicians and researchers who meet these practical criteria. Through the platform, you can explore profiles of registered experts across medical specialties and identify individuals who are actively involved in clinical research or patient care. This makes it easier to find collaborators who are not only knowledgeable but also capable of helping you move from idea to real-world validation.
Structured Early Conversations
Many collaborations fail because important feasibility questions are not discussed early enough. StudyConnect enables founders to book structured conversations with clinicians and domain experts to discuss topics such as clinical endpoints, workflow fit, recruitment feasibility, and potential regulatory hurdles. These early discussions help founders understand whether their product fits real clinical practice and what adjustments may be required before running a study or launching a trial.
Building Multi-Center Networks
Strong collaborations often grow beyond a single site. Through StudyConnect’s network of registered researchers and clinicians, founders can connect with experts who may introduce additional collaborators or institutions. Over time, these relationships can expand into multi-site or multi-center collaborations that strengthen evidence generation and accelerate product validation. By facilitating direct conversations with experienced investigators, StudyConnect helps founders move from isolated discussions to building a broader clinical research network.
Build a Trial-Ready Research Network
One of the most powerful examples of rapid research collaboration came during the early COVID-19 pandemic. Clinicians urgently needed data on how the virus affected cancer patients, and researchers quickly organized a nationwide collaboration across the United Kingdom. Led by Dr. Lennard Lee, the initiative connected 86 cancer centers to collect and analyze real-world patient data. What made this effort successful was not just urgency, but clarity and coordination. The problem was clearly defined, the participating clinicians were already treating the relevant patient population, and the collaboration grew through a network of trusted investigators across institutions. Instead of building the perfect partnership first, they started with capable sites and expanded quickly as momentum built.
This example highlights an important lesson for medical startup founders and research teams: successful collaborations begin with preparation. Before reaching out to potential collaborators, it helps to define a clear one-sentence research question, prepare a concise one-page study brief, outline an authorship and contribution plan, create a shortlist of potential investigators, and verify their trial experience through registries or institutional records. When these elements are ready, outreach conversations become clearer, faster, and more productive.
Ultimately, healthcare collaboration is about execution rather than ideas alone. The strongest research partnerships are built around investigators who have patient access, real clinical trial experience, and sites that are ready to support studies. Starting with one strong collaborator and building outward into a network is often the most effective way to move from concept to real clinical evidence.
If you are building a medical product or planning clinical validation and need help identifying the right experts, StudyConnect can support you throughout the process—from understanding which clinicians you should speak with, to navigating regulatory considerations and gathering early feedback on your product. Our network includes verified clinicians and researchers who regularly work with clinical studies and healthcare innovation. To learn more or begin exploring potential collaborators, feel free to reach out to us at hello@studyconnect.org.




















